Surfactant Content
Non-polar liquids such as toluene, hexane, dodecane, etc, are important for many novel products like electronic paper, batteries, and fuel cells. Dispersions prepared on their basis require stabilization with surfactants. Therefore, formulation and quality control of such systems depends on the ability to monitor surfactant content in the liquid. Conductivity measurements offer such capability. Surfactants, both ionic and non-ionic, enhance the conductivity of non-polar liquids, as shown on Figure below for heptane.
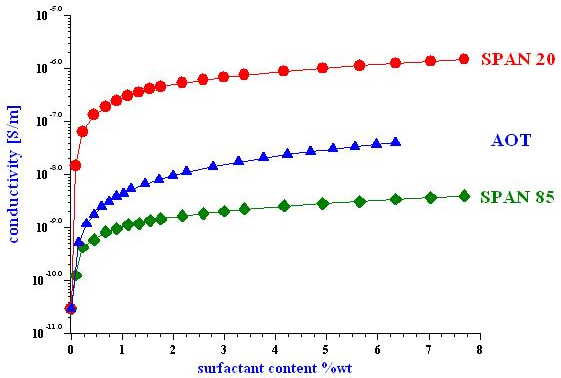
Evidently, a conductivity graph like such serves as a calibration curve for a particular surfactant in a particular liquid. Surfactant content comes as a result of comparing measured conductivity of a sample and then relating it to the surfactant content using the calibration curve. Only with a special conductivity probe capable of measuring conductivities of liquids over a very wide dynamic range, from 10-11 S/m up to 10-4 S/m. Utilization of this simple method can be accomplished
There are two models that can conduct such measurement: standalone non-aqueous conductivity meter DT-700 and Option OP 0041 for DT 1202. Precision of a single measurement is ±(1%+10E-11S/m) over the complete range, and can be statistically improved for multiple measurements.
Application of this instrument for characterizing non-polar liquids and dispersions can be found in several published papers, see for example:
One of the most interesting application of this method is characterization of surfactant adsorption from non-polar liquids into porous materials. Following papers describe this application:
